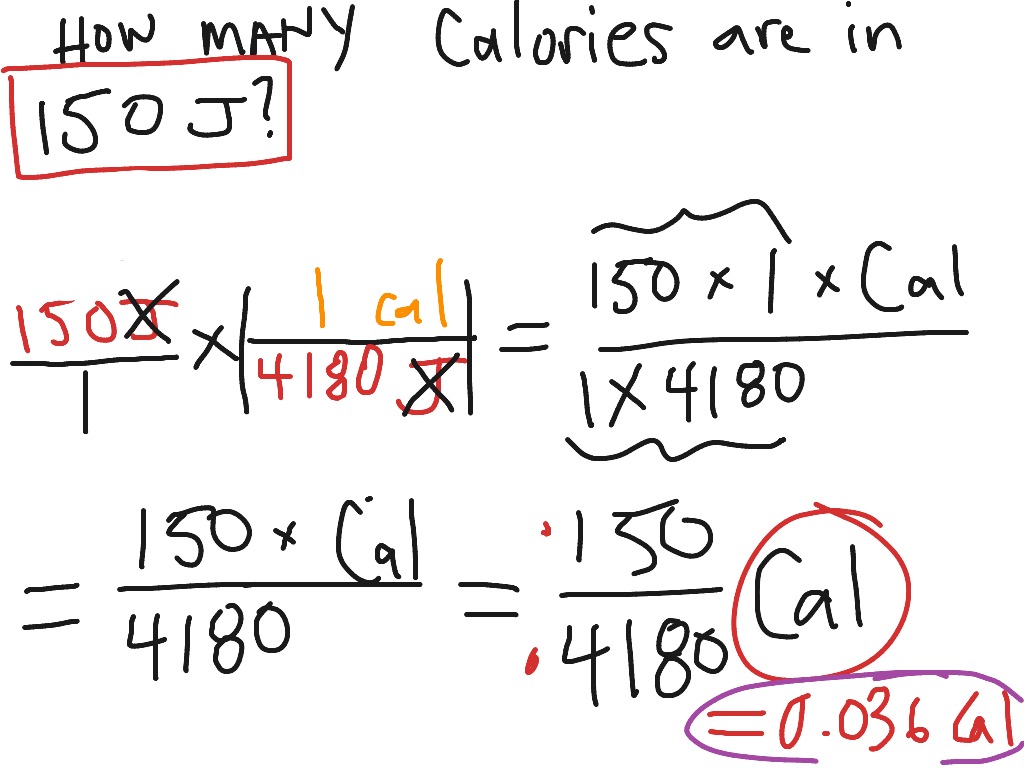69. Energy equivalent to one erg, one calorie andone joule is in the order :(a) 1 Cal>1 Joule>1 Erg(b) 1 Erg>1 Cal>1 Joule(c) 1 Joule>1 Cal>1 Erg(d) 1 Cal>1 Erg>1 Joule | Snapsolve
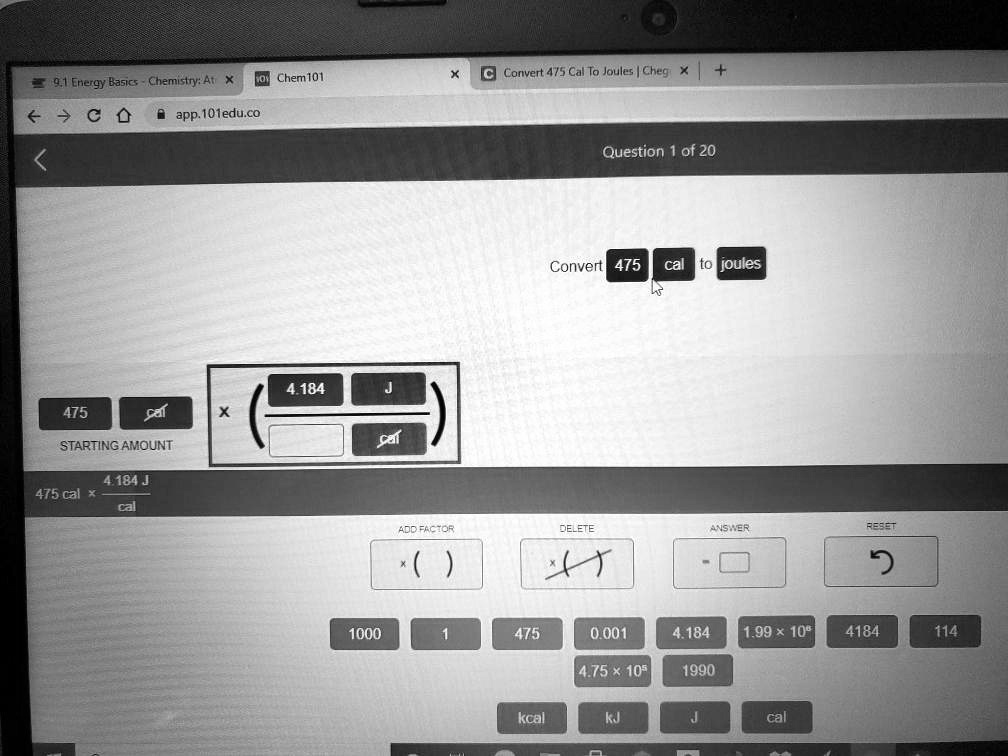
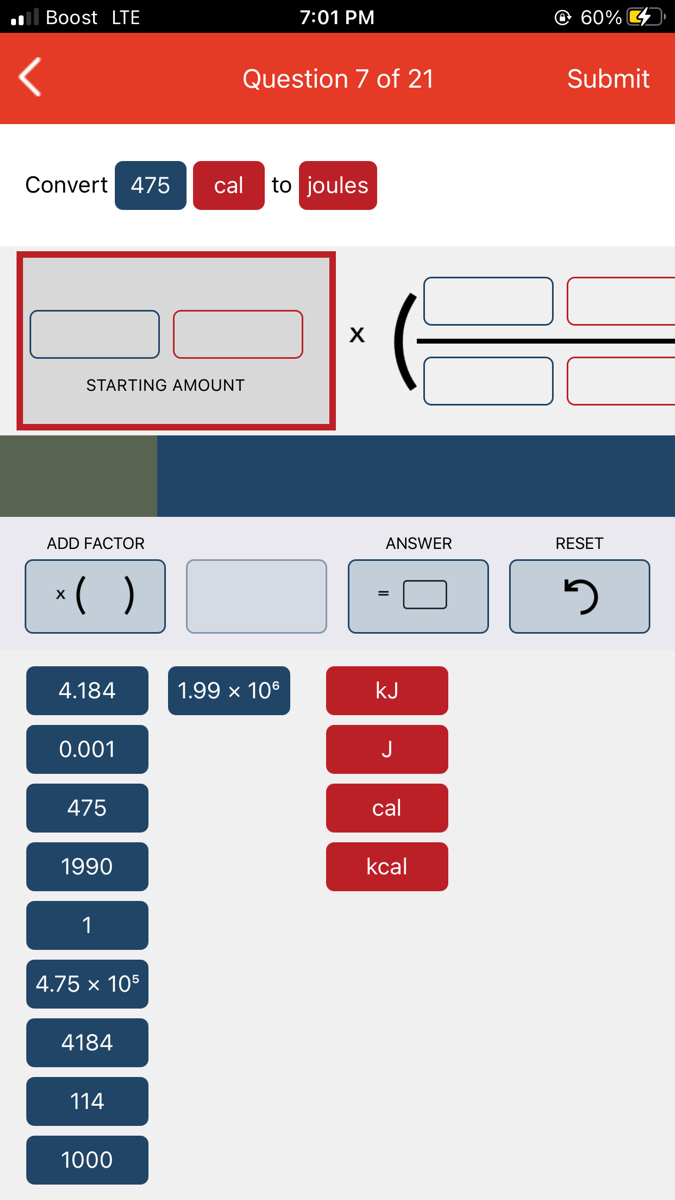
SOLVED:Chem101 Corvert 4/5 Cal To Joules | Chegi 9 1 Energy Basics : Chemistry: Ati app. IOtedu.co Question of 20 Convert 475 to joules 4 184 475 cal STARTING AMOUNT: sal 4.184
Energy Ability to “do work” or produce a change. Forms: potential energy and kinetic energy Types: mechanical, electrical, nuclear, solar, chemical, etc. - ppt download

Energy A Give and Take The Nature of Energy Energy: the ability to do work or produce heat Potential energy (store energy): energy due to position. - ppt download

thermodynamics - Why and who has established that $1\, cal \equiv4.186\, J$? - Physics Stack Exchange

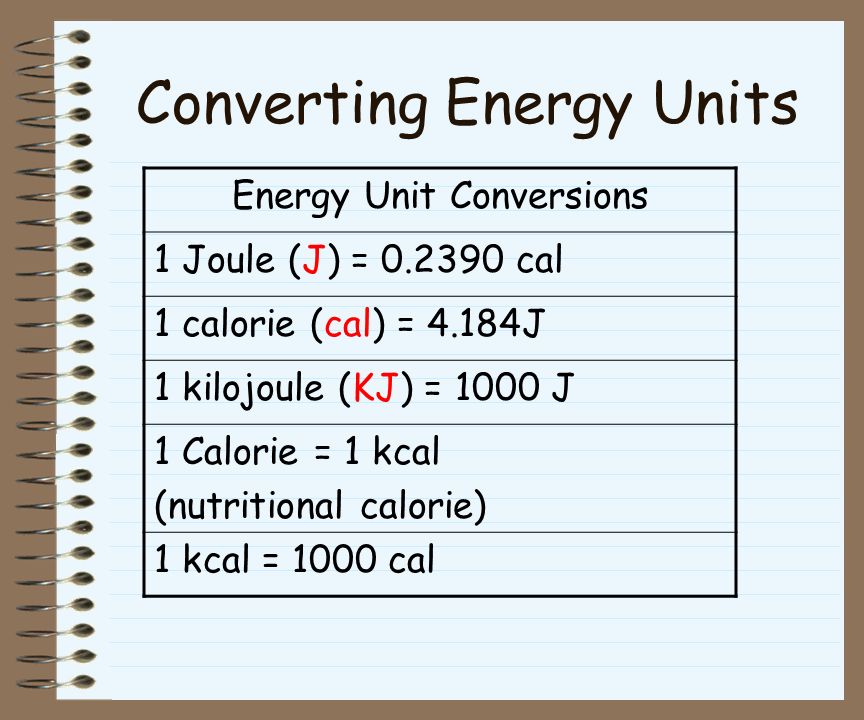
Heat is measured in Joules or calories. 1 cal = J Food energy in measured in Calories (with an upper case C) 1 Cal = 1000 cal. - ppt download

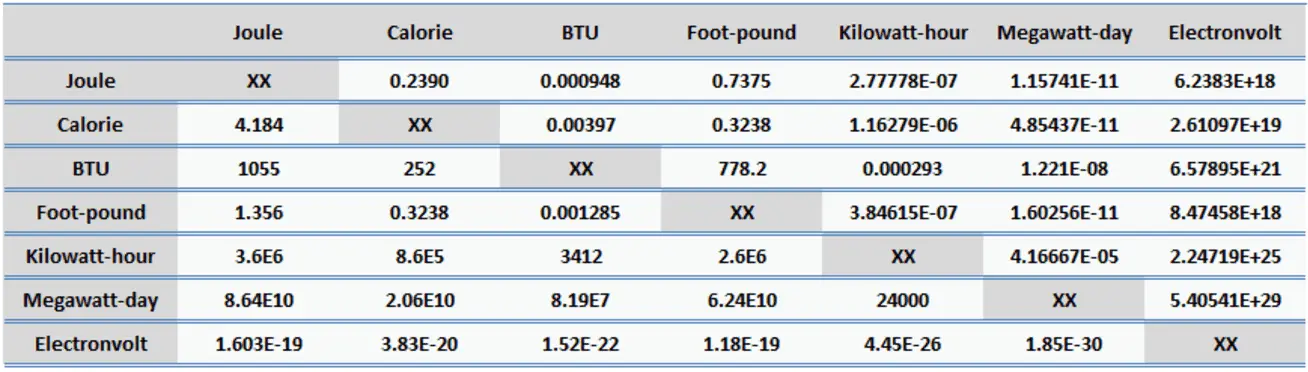
Energy (heat) may be expressed in joules or calories. 1 calorie (cal) = joules (J) How many joules in 60.1 calories? How many calories. - ppt download

SOLVED:01 How much energy in Joules) will it take to raise the temperature of 75.0 g of water from 20.0 to 55.0 0C? ( Specific Heat = 1 cal Ienba ( 3o'
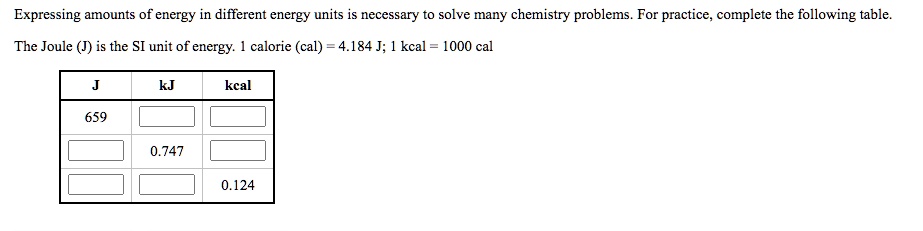
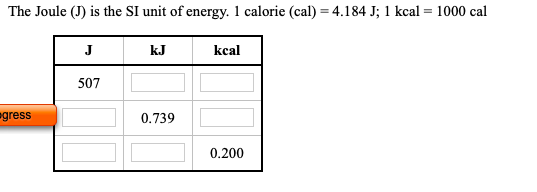
SOLVED:Expressing amounts of energy in different energy units is necessary to solve many chemistry problems. For practice, complete the following table_ The Joule (J) is the SI unit of energy: calorie (cal)

Chapter 10 Energy. 10 | 2 Energy and Energy Changes Energy: ability to do work or produce heat –Chemical, mechanical, thermal, electrical, radiant, sound, - ppt download